Advances in the Treatment of Age-related Macular Degeneration
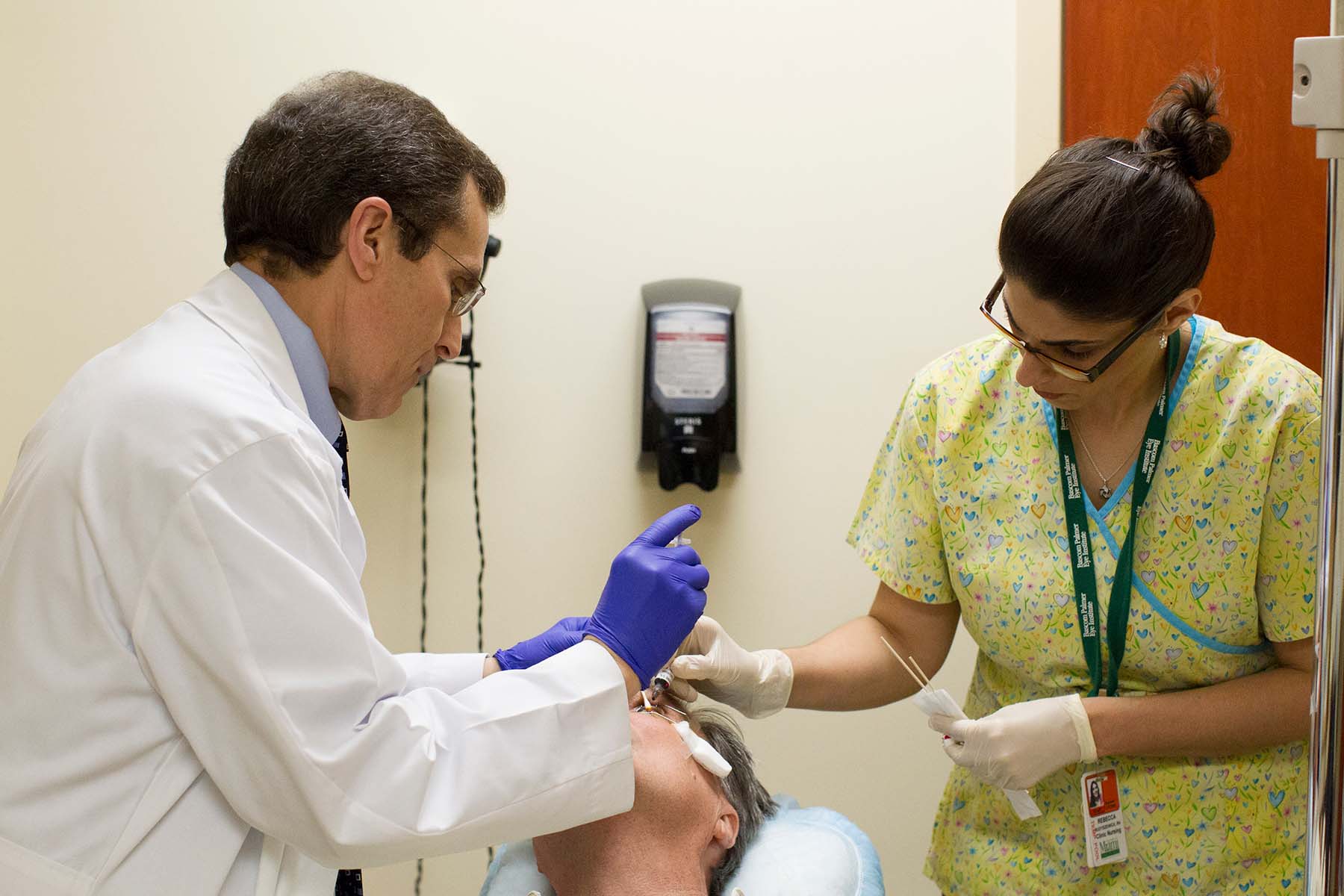
There is a new revolutionary treatment for dry atrophic age-related macular degeneration (AMD) that was approved by the Food and Drug Administration on February 17, 2023. This new drug, which has been in development for the past 18 years, is known as Syfovre®. Syfovre® slows the progression of geographic atrophy in AMD, which is the major cause of blindness from AMD.
AMD, a common cause of vision loss as people age, begins as dry AMD.
As AMD progresses, the dry form of AMD progresses to the late-stage, known as geographic atrophy, or to the late-stage of AMD known as wet AMD.
Wet AMD arises from dry AMD and occurs when abnormal blood vessels grow and leak fluid and blood into the central macula. Only about 15% of patients with dry AMD ever progress to wet AMD.
Since 2005, we’ve had great treatments for wet AMD, starting with Avastin®, followed by Lucentis®, then Eylea®, and now Vabysmo®. All these treatments are highly effective and convert the wet AMD back to dry AMD, but there has been no treatment for dry AMD until Syfovre® was approved. Now the relentless progression of vision loss from geographic atrophy in dry AMD can be slowed and eventually stopped.
We need our central vision for driving, watching TV, seeing people’s faces, and other vital tasks.
The macula is the most important part of the retina because it is responsible for everyone’s best vision. Without treatment, dry AMD will eventually cause vision loss and legal blindness.
How is geographic atrophy treated?
As AMD progresses, the disease eventually causes geographic atrophy to form, which is the loss of retinal tissue that causes the relentless progression of vision loss. Even before legal blindness occurs, patients with early geographic atrophy can still have good reading vision, but they are aware of significant symptoms such as needing brighter lights to read, having difficulty with glare, losing the ability to drive at night, and complaining that they can’t recognize people in dim light.
While Syfovre® isn’t a cure, the drug will slow down the overall vision loss and allow patients to have useful vision for longer resulting in a better quality of life.
Syfovre® won’t improve vision, but it will gradually slow and hopefully stop the progression. However, there is a significant treatment burden. For the first two years, most patients will need monthly injections of Syfovre® into their eyes.
However, the eye must first qualify for treatment.
To know if your eye qualifies, patients need to be evaluated by a retina specialist. While injections into eyes are routinely performed for wet AMD and everyone is nervous when they get their first injections, the procedure quickly becomes less stressful and well tolerated. The procedure performed by the retina specialist is the same as for wet AMD, but the drug we inject is different. Most patients do experience mild discomfort after the injection, and this discomfort lasts for about one day.
Other promising drugs are also being developed for dry AMD, so I’m optimistic that as we treat earlier in the disease, we’ll be able to prevent blindness in almost all our patients with dry AMD.
To schedule an appointment at Bascom Palmer Eye Institute, call 800-329-7000 or click here.

Written by
Philip J. Rosenfeld, M.D., Ph.D., Board-certified Ophthalmologist and Professor of Ophthalmology, Bascom Palmer Eye Institute
Originally published on: March 21, 2022
Tags: AMD, Bascom Palmer Eye Institute, Dr. Philip J. Rosenfeld
