When Standard Cancer Therapy Isn’t Enough: Phase 1 Clinical Trials
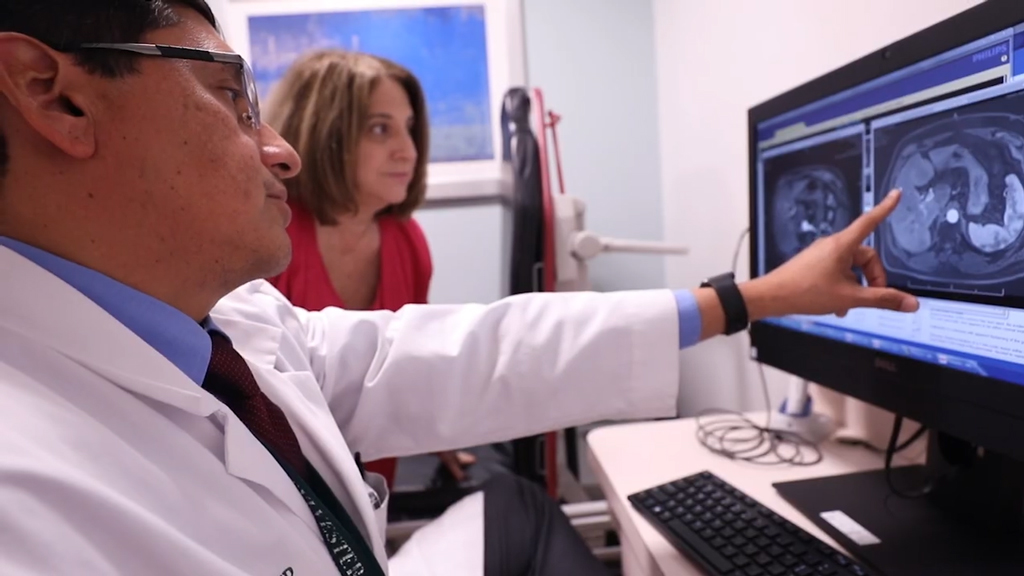
For certain cancer patients, they may be the best choice, says Dr. Jaime Merchan, director of a new Phase 1 program at Sylvester Comprehensive Cancer Center. “We are evaluating drugs never before used in patients and making a difference.”
Clinical trials play an important role in the fight against cancer. Many people with cancer are now living longer because treatments are more effective due to clinical trials. Deciding to participate in a clinical trial allows you to play in important role in advancing scientific knowledge and helping future patients.
The program at Sylvester brings the latest technology and more options to patients for whom standard care for their cancer is not available or there is something complex about their disease or the chemotherapy.
For some patients, the medicines are even helping them control their tumors, so early participation in Phase 1 trials can be pivotal to their treatment.
“I had stage four cancer and wouldn’t be here without this drug,” says Dr. Raymond Mathews, a participant in a Phase 1 clinical trial for kidney cancer. “I take it every day and have no side effects whatsoever.”
Another patient, Luis Martinez, was in remission with urothelial cancer but it came back. In September 2018 he started a phase 1 clinical trial. “I am taking a chance because they are very careful with their investigation. This clinical trial has given me back my life and has meant a new life for me,” he said.
The first studies performed in humans are phase 1 trials. As a result, they are an important step to transitioning novel treatments from the laboratory to the bedside, and often translate into more treatment choices for patients.
Why should I participate?
- Access to promising drugs, medical devices, or treatment approaches before these are available as standard care options
- Free or subsidized cancer treatment for the duration of the trial
- A more active role in your own health care
- Expert medical care at a leading cancer center
- Close monitoring of your treatment and side effects
- The opportunity to make a difference and improve treatments for future generations
The Phase I Clinical Trials Program at Sylvester was created to fulfill the NIH (National Institutes of Health) philosophy for medical research to provide “the necessary foundation for advancing basic and clinical research” to speed promising therapies from the laboratory into the clinic for patient care, all within a safe environment. Notably, it is South Florida’s only academic Phase I testing center dedicated to drug development for cancer patients. For the youngest cancer patients, Sylvester also has Alex’s place, the only Pediatric Phase I Clinical Trials Program in South Florida.
A multidisciplinary team of physicians, scientists, pharmacists, laboratory technicians, nurses, statisticians, research coordinators, and data managers work together to provide the highest standard of care to patients who participate in Phase I trials. The clinic has a dedicated laboratory, pharmacy, and chemotherapy administration areas.
Visit Sylvester.org for more information.
Mary Jo Blackwood, RN, MPH, is a contributing writer for UMiami Health News. Based in St. Louis, MO, and Colorado, she has written medical articles and webpages for consumer publications and major university health centers.
Tags: cancer clinical trials, cancer treatment, clinical trials, Dr. Jaime Merchan, Phase 1 clinical trials
